|
Oxygen is an essential element for life on Earth, as it is required for respiration and combustion. It is a highly reactive element that tends to form anions in ionic compounds and oxides in covalent compounds. In conclusion, oxygen is a non-metal due to its electronic configuration, physical properties, and chemical properties. For example, water is a liquid at room temperature and has a high boiling point due to hydrogen bonding, which is a characteristic of non-metals. 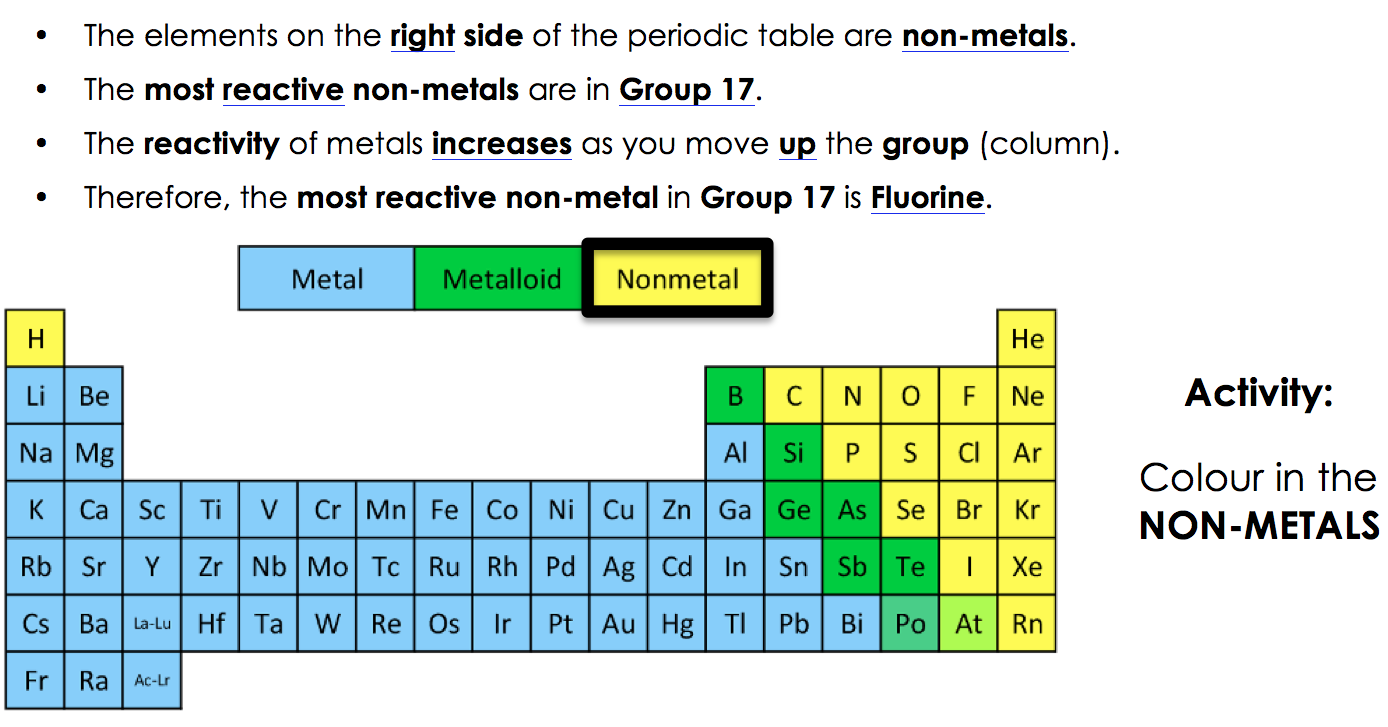
These compounds are typically non-metallic and have properties different from those of their constituent elements. It tends to combine with other elements to form oxides, such as water (H2O), carbon dioxide (CO2), and rust (Fe2O3). Oxygen is highly reactive and can participate in many chemical reactions. 25 Mn Manganese 54.938 26 Fe Iron 55.845 27 Co Cobalt 58.933 28 Ni. Oxygen is colorless, odorless, and tasteless, which makes it difficult to detect without special instruments. It has a low boiling point (-183 ☌) and a low melting point (-218 ☌), which means it is not a solid at room temperature. Oxygen is a gas at room temperature and standard pressure, which is also typical of non-metals. Instead, oxygen tends to gain electrons and form anions (negatively charged ions) in ionic compounds. Occurrence Description 1 Hydrogen H 1 1 s Gas Primordials Non-metal 2 Helium He 18 1 s Gas Primordial Noble gas 3 Lithium. List of elements Atomic Number Name Symbol Group Period Number Block State at. These electrons are negatively charged and tend to repel each other, making it difficult for oxygen to lose electrons and form cations (positively charged ions). This is a list of elements by atomic number with symbol. Oxygen has six valence electrons in its outer shell. Here are some explanations about why oxygen is a non-metal. 
Oxygen is a highly reactive non-metal, which means it does not have metallic properties like malleability, ductility, and conductivity. It is a member of the chalcogen group on the periodic table, which also includes sulfur, selenium, and tellurium. Table 3.1 Element Symbol Type ofHardness Malleability Ductility Conducts Sonority surface Electricity On the bases of the observations recorded in Table 3. The chemical elements can be broadly divided into metals, metalloids, and nonmetals according to their shared physical and chemical properties. Compile your observations regarding metals and non-metals in Table 3.1. Oxygen is a chemical element with the symbol O and atomic number 8. n Carry out the Activities 3.1 to 3.4 and 3.6 with these non-metals and record your observations.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
